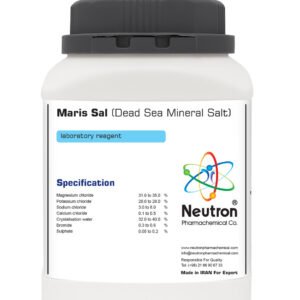
Iron oxide black color
Iron Oxide Black (CI 77499) is a synthetic inorganic pigment with the chemical formula Fe₃O₄, commonly known as magnetite. It is widely used in pharmaceutical and laboratory applications due to its stability, non-toxicity, and excellent coloring properties.
🏭⚗️ Production
Iron oxide black is synthesized through controlled oxidation of iron salts or by precipitation from iron solutions. The resulting pigment is purified to meet the stringent quality standards set by pharmacopeias such as BP, EP, and USP. These standards ensure the absence of toxic impurities like lead and arsenic, making the pigment suitable for pharmaceutical use.
🔬 Properties
Iron oxide black is a fine, dark powder with a characteristic black color. It is insoluble in water and most organic solvents but can be dispersed in oils and certain solvents. The pigment exhibits excellent lightfastness, chemical stability, and heat resistance, making it ideal for use in various formulations. Its magnetic properties are also notable, though they are not typically utilized in pharmaceutical applications.
🧪 Applications
In pharmaceutical formulations, iron oxide black is used as a colorant in tablets, capsules, and coatings to provide a distinctive black color. It is also employed in the production of injectable formulations and topical preparations where color identification is essential. In laboratory settings, the pigment serves as a standard reference for color matching and quality control in the manufacturing of pharmaceutical products.
⚠️ Safety
Iron oxide black is generally recognized as safe for pharmaceutical and laboratory use when produced to pharmacopeial standards. It is non-toxic and poses minimal risk upon ingestion or skin contact. However, as with all fine powders, inhalation should be avoided to prevent respiratory irritation. The pigment should be handled in accordance with standard safety protocols, including the use of personal protective equipment such as gloves and dust masks.